Calcium oxalate- (CaOx) and struvite‐containing uroliths comprise the majority of all urolith submissions from dogs (Box 1). Removal of uroliths will not alter the underlying conditions responsible for their formation, and additional strategies are needed to prevent or minimise urolith recurrence. The most effective prevention strategies are those that eliminate the underlying cause, but in cases where this cannot be identified or eliminated, nutritional support can play a key role. Nutritional support helps to adjust the urine environment, creating a more unfavourable environment for further uroliths to form.
This article explores the nutritional management of canine struvite and calcium oxalate uroliths, focusing on the role of diet in both the dissolution of struvite uroliths and the reduction of recurrence of struvite and calcium oxalate uroliths.
|
Struvite uroliths
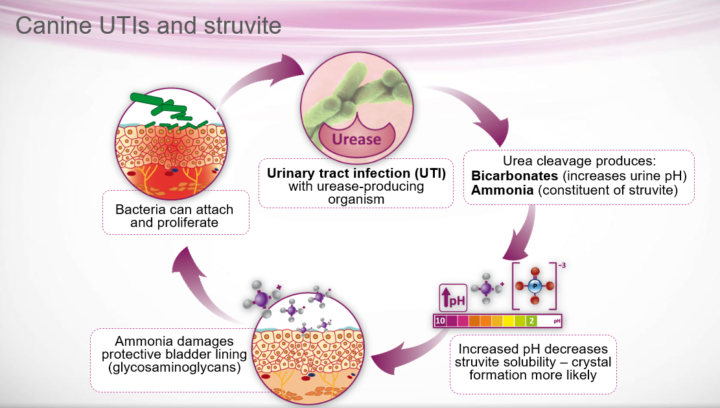
Almost all canine struvite uroliths are infection-induced due to the presence of urease-producing bacteria, usually Staphylococcus pseudintermedius or, less commonly, Proteus mirabilis and Klebsiella species (Dear et al., 2019). These bacteria hydrolyse urea to form ammonia and carbon dioxide, resulting in increased urine pH, which liberates ammonium to form magnesium ammonium phosphate (struvite) crystals (Figure 1).
The majority of canine struvite urolithiasis occurs in females due to host factors that increase the likelihood of bacterial urinary tract infections (UTIs). Note that most Escherichia coli strains do not produce urease, and in patients with non-urease-producing organisms, there is an increased likelihood that they have a different type of stone with a superimposed infection.
In patients with non-urease-producing organisms, there is an increased likelihood that they have a different type of stone with a superimposed infection
Dissolution
Medical management and antibiotics
In all but exceptional cases, medical management with the dissolution of uroliths suspected to be struvite (both sterile and infection-induced struvite uroliths) should be attempted (Lulich et al., 2016). This protocol involves a combination of feeding an appropriate dissolution food with antibiotics. Exceptions can include patient factors (cases where medications or the dissolution food cannot be administered), obstructing uroliths or very large solitary cystoliths where the urolith cannot be adequately bathed in modified urine (Lulich et al., 2016).
Medical dissolution can be highly effective, tends to be less expensive than surgical cystotomy and avoids the risks and complications of anaesthesia and surgery (Lulich et al., 2016). It is considered safe and has not been demonstrated to increase the risk of urethral obstruction (Lulich et al., 2016). Medical dissolution avoids the risk of bladder suture-induced urolith recurrence after cystotomy, which may be responsible for up to 9 percent of urolith recurrences (Appel et al., 2008), and the risk of incomplete urolith removal.
Medical dissolution avoids the risk of bladder suture-induced urolith recurrence after cystotomy, which may be responsible for up to 9 percent of urolith recurrences
Antibiotics are an essential part of therapy for struvite stones and should be based on urine culture and sensitivity results to ensure maximal efficacy. Since struvite stones form in the presence of infection, they trap bacteria within the layers of the stone, and these will be liberated as the stone dissolves. It is, therefore, imperative that antibiotics are continued throughout the dissolution process and for four weeks after radiographic resolution of the urolith(s) (Dear et al., 2019). If antibiotic therapy is discontinued prematurely, infection is likely to recrudesce and inhibit further urolith dissolution.
Diet and fluid intake
There are several commercially available struvite dissolution diets available on the veterinary market, some of which have clinical studies to demonstrate their efficacy. Feeding these specially designed functional diets can be very effective to achieve full struvite dissolution in as little as three weeks for both sterile and infection-induced struvite urolithiasis, although it may take over three months in some cases (Bartges et al., 1992; Dear et al., 2019). The diet should be fed exclusively throughout the dissolution process – something important to emphasise to pet owners. These diets promote the formation of acidic urine (pH below 6.5), significantly increasing struvite solubility. They are also reduced in phosphorus and magnesium (struvite urolith precursors), helping to promote struvite dissolution. In some cases, they may also be reduced in protein which helps reduce urea, resulting in loss of the renal medullary concentrating gradient and helping to promote diuresis.
Promoting fluid intake is also critical to help reduce the concentrations of urinary calculogenic minerals and increase crystal evacuation
Promoting fluid intake is also critical to help reduce the concentrations of urinary calculogenic minerals and increase crystal evacuation. Methods include increasing drinking water and increasing moisture in the diet, for example by feeding a canned diet or, if not possible, a dry diet soaked with water, although the efficacy of this latter recommendation has not been formally evaluated (Dear et al., 2019). A target urine specific gravity (USG) of 1.020 or below should be aimed for and monitored throughout the dissolution process.
Monitoring
Urinary pH should also be monitored, ideally with a pH meter (since these are more accurate than urinary dipsticks). Target urinary pH can differ between dissolution diets, so attention should be paid to the targets published by the manufacturer for the specific diet chosen. Urine sampling should be done just prior to a meal or over eight hours after meals to minimise confounding post-prandial alkalinuria. In a minority of cases, if urine remains persistently alkaline despite exclusive feeding of the dissolution diet and administration of appropriate antibiotics, dl-methionine or ammonium chloride may also be required to acidify the urine (Bartges and Moyers, 2010). This is because struvite solubility is greatly increased by acidifying the urine (pH below 6.5).
Alongside urinalysis, the percentage of dissolution should be regularly evaluated by radiographs (for example, every two to three weeks). Potential reasons for minimal reduction in stone size or unsuccessful full dissolution are detailed in Table 1.
| Poor compliance with the diet (by pet or owner) | Insufficient increase in fluid intake | Inadequate antibiotic therapy | Urolith size or burden | Wrong stone type | Composite stone |
|---|---|---|---|---|---|
| This may include feeding an alternative diet or feeding additional foods alongside the dissolution diet Poor compliance can be monitored with urinalysis in most cases. Manufacturers will publish factors such as target urinary pH produced by their diets | USG of 1.020 or below should be aimed for, although dissolution can still be achieved above this value (Dear et al., 2019) If urine remains very concentrated, more aggressive methods to increase fluid intake should be instituted, eg adding additional moisture to a canned diet | Antibiotics should be selected based on culture and sensitivity to avoid potentially ineffective treatment due to antimicrobial resistance Duration of antibiotic therapy should be considered, and antibiotics should be given throughout the dissolution trial and continued for several weeks past radiographic resolution Poor client or pet compliance may result in an insufficient dose or regularity of antibiotic administration | If, for example, the urolith is too large to be bathed in modified urine, dissolution may be ineffective (Dear et al., 2019) | If there is no decrease in urolith size during the dissolution trial despite previous factors being ruled out, the stone may be of another mineral type | If there is a decrease in urolith size but full dissolution is not achieved, consider a composite stone with a non-struvite component. For example, calcium phosphate can be seen in association with a urinary tract infection (Dear et al., 2019) |
Prevention of recurrence
Since struvite stones rarely form in the absence of an infection, they can be prevented by managing risk factors for urinary tract infections to eliminate their occurrence. You can also routinely screen for infections to enable early detection and treatment.
Urine sediment evaluation and pH monitoring are not suitable diagnostic substitutes for aerobic bacterial urine cultures (Lulich et al., 2016). A long-term urinary diet is not necessarily required and plays only a minor role. It will not prevent the recurrence of struvite uroliths, although it may delay or minimise the urolith burden in the presence of an unrecognised UTI (Lulich et al., 2016).
In contrast, in the case of sterile struvite uroliths (though rare in dogs), nutritional prevention plays an essential role (Lulich et al., 2016). Most diets appropriate for dissolution, which acidify the urine and contain low magnesium and phosphorus, are also appropriate for long-term feeding. Wherever possible, feeding a diet high in moisture to reduce urine specific gravity is also a cornerstone of management, as it is for almost all uroliths (Lulich et al., 2016).
Calcium oxalate uroliths
CaOx uroliths cannot be medically dissolved and require mechanical removal (for example, by cystotomy, voiding urohydropropulsion or endoscopic laser lithotripsy). However, nutritional support can be used to reduce the risk of stone recurrence after removal and thus plays a crucial role since CaOx is a highly recurrent urolith, with approximately half of dogs re-developing CaOx uroliths within two years.
The factors influencing the risk of CaOx uroliths are not well understood, making it a challenging stone to prevent. Genetic factors are believed to play a role, and many dogs with CaOx uroliths have increased urinary calcium excretion despite normal blood calcium (ie idiopathic hypercalciuria) (Furrow et al., 2015). However, serum ionised and total calcium concentrations should be measured in all patients; if elevated, this may increase hypercalciuria. Controlling hypercalcaemia is essential to the long-term prevention of CaOx urolith recurrence, and further diagnostic testing is likely to be warranted if it is identified. Sex also influences risk, with CaOx stones more common in male dogs. Underlying medical conditions can significantly increase risk; for example, hyperadrenocorticism (HAC) increases the risk of calcium-containing uroliths forming up to ten-fold compared to dogs without HAC (Hess et al., 1998), although the mechanism is not understood.

Prevention
Prevention strategies are based on the physiology of calcium and oxalate excretion, surrogate endpoints (eg relative supersaturation (RSS) (Figure 2)) and identification of risk factors. However, these do have limitations. For example, RSS is a mathematical model that may not correlate well with calcium oxalate urolith formation (Lulich et al., 2016).
Crystal growth and, possibly, initial crystal formation are at least partly a reflection of urine supersaturation. So, controlling the risk factors promoting urine CaOx supersaturation (such as hypercalciuria, hyperoxaluria and highly concentrated urine) should help minimise urolith recurrence. Increasing water intake is an essential component of management.
Diet
Selecting the most effective diet to minimise the risk of recurrence is challenging because appropriate studies (prospective case-controlled studies of three or more years) evaluating recurrence have not yet been performed.
High-moisture (for example, canned) foods containing over 75 percent water should be recommended wherever possible to maximise urine volume and reduce urine concentration. Most available commercial diets that minimise the risk of CaOx recurrence offer a wet form. For patients refusing a wet diet, an alternative option could be the addition of sufficient water to dry kibble to significantly increase moisture intake. Monitoring of urine specific gravity should be conducted regularly, with a target of 1.020 or below.
Acidification of the urine (pH below 6.5) should be avoided, as it can increase hypercalciuria, and most diets designed to minimise CaOx urolith recurrence are neutralising. While CaOx is less pH-dependent than struvite, diets promoting acidic urine in dogs have been associated with CaOx urolith formation (Okafor et al., 2014).
If urine pH persistently remains below 6.5 despite exclusive feeding of an alkalinising diet, additional oral administration of potassium citrate […] can be considered
The ingestion of foods containing high quantities of animal protein (over 10g/100kcal) contributes to calcium oxalate uroliths by promoting acidic urine, increasing urine calcium excretion and decreasing urine citrate excretion. Lower protein levels may promote diuresis and minimise the calcium and oxalate concentrations in the urine.
Commercial diets designed to minimise CaOx recurrence also tend to have reduced oxalate concentrations and a moderate but not excessive restriction of calcium. Very low calcium concentrations are not recommended, as this can increase oxalate absorption. Potassium citrate may be included, which is helpful as citrate chelates calcium, which lowers supersaturation and inhibits crystal formation. Potassium citrate will also alkalinise the urine. If urine pH persistently remains below 6.5 despite exclusive feeding of an alkalinising diet, additional oral administration of potassium citrate (at a dose of 150mg/kg/day) can be considered to promote a more neutral pH (Lulich et al., 2016).
Sodium supplementation
There is a lack of consensus on whether sodium should be supplemented to prevent CaOx uroliths. High-sodium dry foods should not be recommended as a substitute for high-moisture foods (although could be considered if the owner declines a wet (canned) diet). Sodium supplementation can increase water intake, thus diluting urinary calcium and oxalate; however, these effects may be short-lived. Sodium decreases urinary calcium resorption by the kidneys, resulting in increased calcium excretion. In short-term feeding trials in dogs, this effect appears to be cancelled out by the increased urine volume (Lulich et al., 2005), but long-term studies evaluating the efficacy and safety of dietary supplementation with sodium chloride in dogs with CaOx urolithiasis have not been conducted therefore supplementation should only be recommended cautiously (Lulich et al., 2005).
Minimising recurrence
The high recurrence rate of CaOx uroliths warrants a comprehensive approach and regular monitoring to ensure target USG and urinary pH are being achieved and scant or no CaOx crystals are visible on microscopy. Diets designed to minimise the recurrence of calcium oxalate uroliths should be fed exclusively. Clinicians should recommend a diet with PARNUT (foodstuffs for particular nutritional uses) and ensure that owners do not select a grocery diet. This is because some of these may make claims about maintaining urinary health but will not be adequately formulated for urolith management.
The high recurrence rate of CaOx uroliths warrants a comprehensive approach and regular monitoring to ensure target USG and urinary pH are being achieved
Vitamin C supplements should be avoided as they may increase oxalate excretion, as should any supplements containing cranberry, since these have been associated with increased urinary oxalate excretion in humans (Redmond et al., 2019). Owners should be cautioned to avoid foods containing high levels of calcium, such as milk and cheese, and high oxalate foods should be avoided. These include, in particular, nuts – including peanut butter – and vegetables such as spinach, as well as human foods such as chocolate and rhubarb.
In addition to the above nutritional recommendations, thiazide diuretics may be required in patients with highly recurrent calcium oxalate uroliths. These enhance renal tubular reabsorption of filtered calcium and may indirectly affect intestinal calcium absorption and bone calcium deposition. A 55 percent decrease in urinary calcium concentration was reported in urolith-forming dogs administered 2mg/kg hydrochlorothiazide twice daily (Lulich et al., 2001), although long-term controlled clinical trials are needed to confirm the safety and effectiveness.
Conclusion
Struvite and calcium oxalate uroliths have different risk factors, and management differs significantly during the initial treatment of any uroliths identified and when minimising the risk of recurrence. However, nutritional support plays a valuable role in managing both stone types, usually considering a combined approach of adjusting the urine pH, increasing urinary volume and altering dietary components. Regular urinalysis and other monitoring strategies, including imaging, should be conducted to ensure management strategies employed are effective.
Different key nutritional factors have been identified in the management of each stone type and incorporated into several commercially available diets designed to manage struvite and/or calcium oxalate uroliths. Some manufacturers produce dual diets designed to support both urolith types, whereas others produce separate products. Attention should also be paid to supplements and extras as, wherever possible, it is usually desirable to feed a urinary diet exclusively to maximise the likelihood of success – whether this is dissolution or prevention of recurrence (dependent on stone type) – and owners should be educated about this. Maximising fluid intake is also a cornerstone of management.











