Traumatic brain injury (TBI) can occur in many different cases; it has been reported that up to 20 percent of patients seen in the emergency veterinary clinic will present with TBI. More often than not, these patients will have been involved in a collision and have other concurrent disease processes that may affect the outcome of the TBI. When managing these cases, it is crucial to consider these patients as a whole, thinking about how each concurrent disease process (eg hypovolaemia) and its treatment will affect the factors influencing TBI outcomes.
Veterinary professionals must be able to recognise and manage the signs of TBI to prevent further injury. In human medicine, several guidelines have been developed to help maintain cerebral perfusion – a key factor to successful outcomes in head trauma cases.
What do we need to know about cerebral perfusion pressure, cerebral blood flow and intracranial pressure in cases of traumatic brain injury?
Cerebral perfusion pressure (CPP) is the net perfusion pressure that drives oxygen into the cerebral tissues. It is primarily dependent on the mean arterial pressure (MAP) and is the difference between MAP and intracranial pressure (ICP).
CPP is important because of the significant part it plays in maintaining cerebral blood flow (CBF) and cardiovascular resistance, which is often affected in motor vehicle accidents. Autoregulation is triggered when there is increased blood pressure and cerebral vascular resistance. When triggered it causes cerebral vasoconstriction and a decrease in CBF, thus resulting in less cerebral blood volume, which is responsible for supplying oxygen and glucose, making it critical to ICP homeostasis. This autoregulation is a coping mechanism of the body to maintain acceptable levels of ICP and CPP. Patients with TBI often develop cerebral hyperperfusion in the early stages of injury – this is significant if the cerebral oxygen consumption is not matched.
ICP is the sum of brain parenchyma, cerebral blood volume and cerebrospinal fluid (CSF). As the skull is a rigid structure, there is an element of intracranial compliance that allows fluid exchange and protection. In TBI patients, there is often parenchymal damage and blood leakage, causing an exceedance of intracranial compliance capacity and, therefore, raised ICP and, consequently, raised CPP. The body regulates ICP by shunting CSF and blood to the body and away from the brain, as raised intracranial hypertension will cause ischaemia over time.
Pathophysiology of traumatic brain injury
Primary TBI is a result of a direct mechanical force and will cause concussion, contusion (producing parenchymal haemorrhage and oedema), laceration and extra-axial haemorrhage on impact. Limitation of the initial injury is often futile, but the aim of treatment is to reduce the likelihood of secondary TBI by avoiding hypoxia and hypoperfusion. Hypoxia of the tissues and hypoperfusion cause lactate accumulation, leading to ischaemia, wherein ATP stores are depleted, preventing the repair of tissues. Raised ICP will follow this cascade in reaction to hypoxia and hypoperfusion, leading to further active haemorrhage and compromise of the blood–brain barrier.
Limitation of the initial injury is often futile, but the aim of treatment is to reduce the likelihood of secondary TBI by avoiding hypoxia and hypoperfusion
Secondary TBI refers to a cascade of pathophysiological processes that lead to “delayed non-mechanical” neuronal damage, both locally and distally to the primary injury. It is characterised by terminal membrane depolarisation, leading to the excessive release of neurotransmitters (glutamate and aspartate). These excitatory neurotransmitters activate N-methyl-D-aspartate receptors as well as voltage-dependant calcium-sodium channels, repeatedly causing a catabolic intracellular process, which, in turn, releases free radicals and fatty acids. The release of free radicals leads to inhibition of DNA repair and fragmentation, which ultimately leads to necrosis or apoptosis.
Therapy aims to prevent this decreased global oxygen delivery (DO2) by ensuring the cardiac output (CO) as well as the oxygen content of the arterial blood (CaO2) is sufficient. CO is affected by the stroke volume, which in trauma patients may be depleted secondary to blood loss, potential restricted fluid administration and the administration of diuretics, and alterations to oxygen saturation as a result of pleural space disease or pulmonary contusions (often seen in these patients).
Assessing patients with traumatic brain injuries
Heart rate, pulse quality and synchronicity, blood pressure, lung changes, respiratory rate and temperature are key markers for TBI. Electrocardiograms and blood pressure monitoring should be part of the major body assessment. Animal Trauma Triage (ATT), developed in 1994, is a validated scoring system designed to assess six essential categories: perfusion, cardiac, respiratory, eye/muscle/integument, skeletal and neurological. The maximum score a patient can be given is 18, with a higher score indicating increased mortality.
Heart rate, pulse quality and synchronicity, blood pressure, lung changes, respiratory rate and temperature are key markers for traumatic brain injury
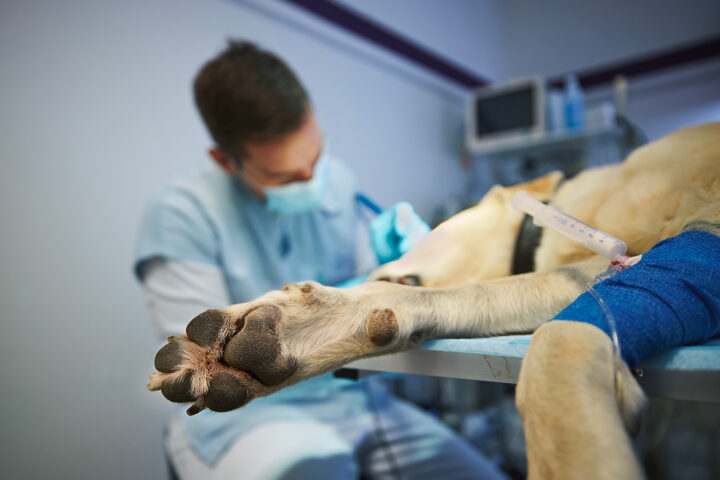
Other markers (such as the clinical signs listed in the next section) can be used in conjunction with ATT and a modified Glasgow Coma Scale (MGCS) (Figure 1) to assess the patient’s motor activity, brain stem reflexes and level of consciousness. These assessments will help assess morbidity and the potential success of treatment. The MGCS has a total score of 18, though unlike the ATT, a lower score indicates a poorer prognosis. This scoring system serves as a serial monitoring tool to indicate improvement and deterioration.
The incidence of TBI can induce the pathways to cause neurological pulmonary oedema (NPE). NPE causes dyspnoea, hypoxaemia and hypercapnia, all of which can lead to an increase in ICP and mortality. The pathophysiology of NPE is thought to be marked peripheral vasoconstriction that leads to systemic and pulmonary hypertension, resulting in oedema.
Clinical signs of secondary traumatic brain injury
Posture
Patients with TBI will often present non-ambulatory, and it is important to consider whether these patients have concurrent spinal fractures. Patients with TBI often present with decerebrate rigidity (Figure 2), decerebellate rigidity and a Schiff-Sherrington posture. Decerebrate rigidity indicates problems in the rostral brainstem and can increase morbidity and further increase decerebellate rigidity.

Pupil size and symmetry
Pupil size is affected by the parasympathetic and sympathetic systems: the parasympathetic light response to bright light will contract the pupils, but sympathetic responses will expand the pupils in darkness. Testing both pupils with the pupillary light reflex test (as well as covering the opposite pupil to see if it has the same response as the uncovered one) can identify the location of the TBI. This is because sympathetic neurones are found in the thalamus, whereas the parasympathetic responses are found in the medulla oblongata.
Cushing’s reflex
Cushing’s reflex, also known as the vasopressor response, is a physiological nervous system response to increased ICP.
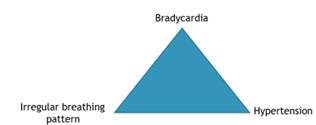
The triad known as the Cushing’s reflex includes increased blood pressure, bradycardia and irregular breathing (Figure 3) and often indicates herniation or severe haemorrhage. If a patient with a suspected TBI is showing any signs of the Cushing’s reflex triad, then treatment must be initiated immediately, though morbidity factors are significantly increased.
If a patient with a suspected TBI is showing any signs of the Cushing’s reflex triad, then treatment must be initiated immediately, though morbidity factors are significantly increased
Hyperthermia
When trauma occurs to the thermoregulatory centre, hyperthermia may be present. (This is also true of iatrogenic disturbances to the thermoregulatory centre, stress, seizures or pain.) Hyperthermia should be addressed immediately to stop increases in cellular metabolism and vasodilation, both of which may result in an increase in ICP.
Treatment and management of traumatic brain injury
Veterinary nurses should always apply the ABCs of triage to TBI patients, as this will often alert the clinician to changes directly affecting secondary TBI. It will also help address any life-threatening issues arising with the three major body systems: the cardiovascular, respiratory and central nervous systems. Treatment for these life-threatening problems should be implemented immediately to ensure that the delivery of oxygen is maximised, as hypovolaemia and hypoxaemia are shown to strongly correlate to increased mortality.
Treatment for these life-threatening problems should be implemented immediately […] as hypovolaemia and hypoxemia are shown to strongly correlate to increased mortality
The treatment of TBI patients can be categorised into three tiers, discussed below.
1) Tier one
Tier one treatment is basic treatment that should be offered to all TBI patients. Oxygenation is the most important treatment for tier one TBI patients, as treatment aims to increase blood oxygenation by maintaining the partial pressure of oxygen (PO2) in the arterial blood as close to normal (80mmHg) as possible. Regulation of a normal PO2 will assist in the autoregulation of CBF, as well as baseline values of the partial pressure of carbon dioxide (PCO2), blood glucose and blood pressure.
Consideration of whether the patient requires mechanical ventilation will depend on the responsiveness to oxygenation, which can be measured via blood gas values. If ventilation is necessary, practitioners should avoid prongs and nasal catheters due to the increased risk of raising ICP.
Blood products may be needed to increase the oxygen carrying capacity (if packed cell volume is less than 20 percent); other products such as oxyglobin can be used to achieve this.
Pain assessments should be carried out following the initial exam. Following this, pain management should be selected carefully as non-steroidal anti-inflammatories (NSAIDs) are contraindicated, and some opioids can cause respiratory depression (hypercapnia) or tachypnoea (hypocapnia) if given rapidly. However, TBI patients will most likely be painful, which can cause both of these ventilation changes.
Fluid therapy is the second mainstay of tier one treatment. It is important to consider the patient’s blood pressure, pulse quality and whether there is shock (TBI patients often suffer multi-focal trauma) and/or some degree of dehydration. Initial resuscitation is often achieved with crystalloid boluses and the use of hypertonic saline to restore circulating volume and blood pressure while limiting the volume of fluid administered.
It should be noted that in tier one management, thermoregulation is important to perfusion (vasoconstriction/vasodilation), and it has been recommended that the TBI patient temperature remains between 37 and 39°C.
2) Tier two
Tier two treatment looks at therapies for TBI patients unresponsive to tier one and progressively deteriorating. Osmotic diuretics fall into this category; however, the patient must be well perfused before starting this treatment due to the risk of dehydration, systemic hypertension and hyperkalaemia.
Tier two treatment looks at therapies for traumatic brain injury patients unresponsive to tier one and progressively deteriorating
Mannitol is classified as an osmotic diuretic; therefore, it is imperative for the patient to have had tier one therapy (fluid therapy) prior to starting this treatment. The main mechanism of action for mannitol is the promotion of vasoconstriction through a reduction in blood viscosity, which, in turn, reduces ICP. It will also reduce oedema by decreasing the production of CSF to create a difference in osmolarity to help drain the extravascular fluid into the intravascular space.
3) Tier three
Tier three and additional therapies include:
- Surgery for fractures, acute extra-axial haematoma and CSF leaks
- Mechanical ventilation to regulate hyperventilation – hypercapnia can cause excessive intracranial vasodilation, leading to increased ICP
- Glucose control
- Seizure control
- Nutritional supplementation
All patients should also receive physical therapy (massage, passive range of movement) to avoid decubitus ulcers. Other crucial therapies include the placement of urinary catheters +/- faecal catheters or enemas to avoid soiling and scalding, lubrication of the eyes and mouth to avoid ulcers and, most importantly, the head and neck should be elevated together at a 20- to 30-degree angle. Jugular catheters and jugular blood sampling should be avoided to prevent compression of the jugular vein (causing increased ICP) and unnecessary movement of the head.










